Customized Medical Blister Trays and Packaging | Sterility and Safety Solutions
Application areas of pharmaceutical plastic trays


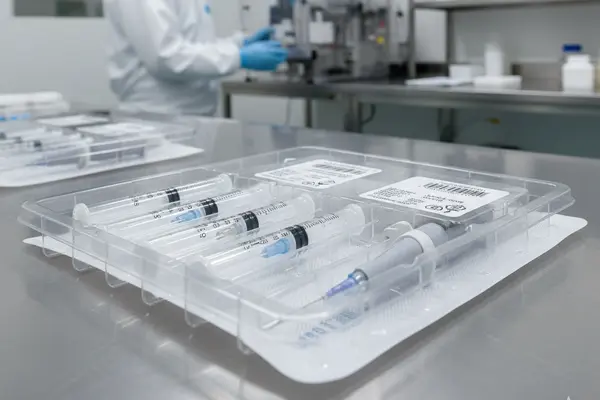
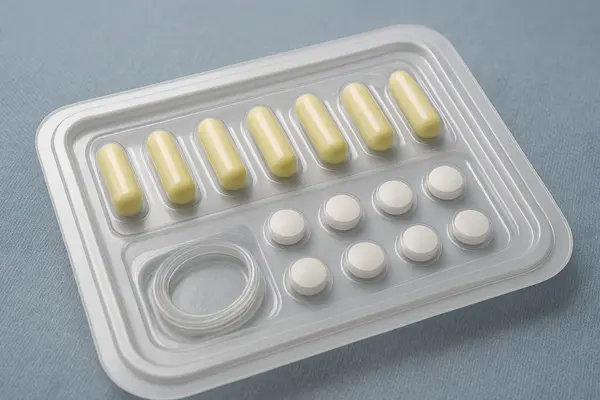
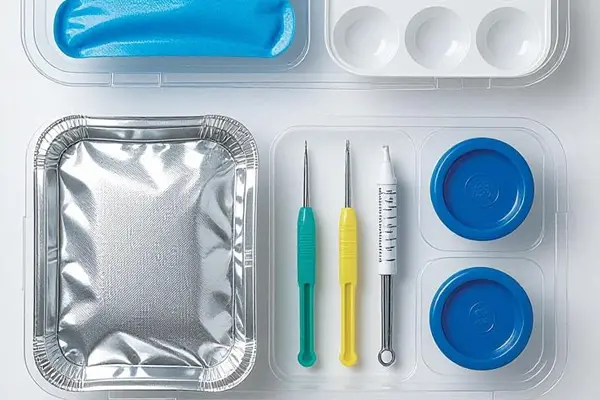



Why choose our Medical blister tray ?
Cleanroom Production Ensures Sterility Compliance
All of our medical blister trays are manufactured and packaged in cleanroom environments certified to ISO 7 or higher standards. From raw materials to finished product, we rigorously monitor for particulate matter and biological contamination, ensuring that the packaging we deliver meets your highest sterility standards.
Superior Sterilization Compatibility and Integrity
Our medical trays, made from specialized materials like PETG or medical-grade HIPS, are designed for compatibility with all major sterilization methods, including ETO (ethylene oxide), gamma radiation, and electron beam sterilization. The packaging maintains its structural integrity during sterilization, effectively maintaining a sterile barrier.
Tight Dimensional Tolerances and Precision Fit
We utilize high-precision molds and advanced thermoforming technology to ensure the tray's exceptional dimensional accuracy. Every groove in the tray fits perfectly with your medical device, minimizing movement and friction, effectively preventing damage to delicate instruments during transport.
Full suite of international certifications for fast validation
We provide all necessary material compliance certificates and verification reports, including USP Class VI testing compatibility, RoHS, and REACH declarations. This helps you quickly pass the product and packaging validation processes required for FDA or CE registration, accelerating your time to market.
Reliable Sterile Barrier System (SBS)
Our tray's sealing flange is designed to maintain strict thickness and width consistency. This ensures a strong seal and uniform peel when heat-sealed with Tyvek® or a medical-grade lidding material. This provides reliable protection against moisture and contamination.
Traceability and Transparent Batch Management
We implement a strict batch control and traceability system, recording all data from raw material batches to final shipment date. If any quality issues arise, we can quickly identify and trace the batch back to its source, providing you with the most reliable supply chain assurance.
Optimizing the operating experience for healthcare professionals
Our trays are designed with user-friendliness in mind, including clear product visibility, an easy-to-peel opening, and a logical layout of recommended procedure packs, helping healthcare professionals perform procedures quickly and accurately.
Long-term, stable supply and risk management
As a manufacturer with advanced production lines, we can provide long-term, stable, and large-scale supply guarantees. We implement a strict dual material sourcing strategy and risk management plan to ensure your packaging supply chain remains uninterrupted in the event of an emergency.
FAQs about Medical Product Blister Trays
What standards must your medical blister tray production environment meet?
Production must typically take place in a certified Cleanroom, usually required to meet ISO 7 or ISO 8 standards. This controls particulate matter and microbial contamination to ensure the product meets sterility requirements.
What are the most common materials used for medical blister trays?
The most common are PETG (offering toughness and clarity), HIPS (economical and impact-resistant), and PP (Polypropylene). Material choice depends on the required sterilization method and product protection needs.
How do you ensure the sterility of the trays?
We control bioburden through Cleanroom production and use materials like Tyvek® lidding to form a Sterile Barrier System (SBS), which maintains the tray’s sterile state after sterilization.
Are your medical trays compatible with common sterilization methods?
Yes. Our material formulations are designed to be compatible with mainstream methods like ETO (Ethylene Oxide), Gamma radiation, and E-beam sterilization, maintaining structural integrity throughout the process.
What is "Peel Strength" for a medical tray?
Peel strength is the force required to separate the lidding (e.g., Tyvek®) from the tray flange. We strictly control this to ensure it provides a reliable seal while allowing healthcare professionals to peel smoothly and cleanly during aseptic procedures.
How do I determine which tray material my medical device needs?
It mainly depends on two factors: the product’s weight/shape (determining the required rigidity) and the sterilization method. We recommend the most suitable medical-grade material based on your device and sterilization needs.
What is the Minimum Order Quantity (MOQ) for medical blister trays?
Due to the need for high-precision custom molds and Cleanroom production, the MOQ is higher than for standard packaging. The specific quantity is determined based on your tray size and annual usage projection.
How do you ensure consistency in tray dimensions during mass production?
We use high-precision steel molds and implement strict in-line optical inspection during production. This ensures that the tray’s depth and dimensional tolerance maintain extremely high consistency across the entire batch, meeting automated assembly and sealing requirements.
Can your medical trays be used for implant packaging?
Yes, we can provide trays that meet the stringent requirements for implant packaging. These trays typically use highly biocompatible medical-grade materials and come with complete material compliance documentation.
What is the difference between Tyvek® and medical paper as lidding?
Tyvek® offers superior microbial barrier and puncture resistance, making it the preferred choice for most sterilization methods. Medical paper is lower cost and often used for steam sterilization, but it provides a less robust barrier than Tyvek®.
Have Anything To Ask Us?
Please fill in your email in the form and we’ll get back to assist you soon!
